2P Holography | Maximum Power
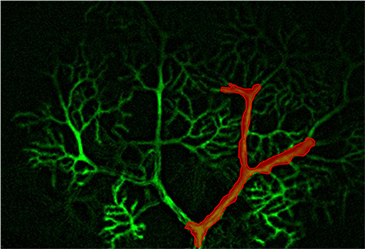
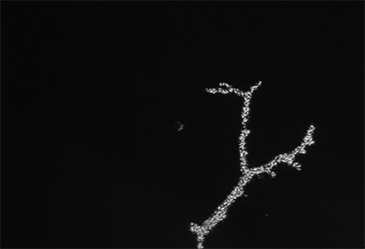
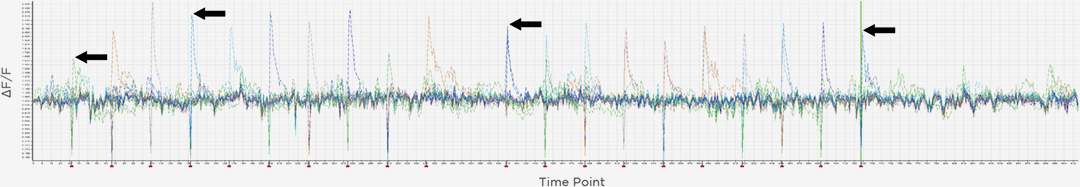
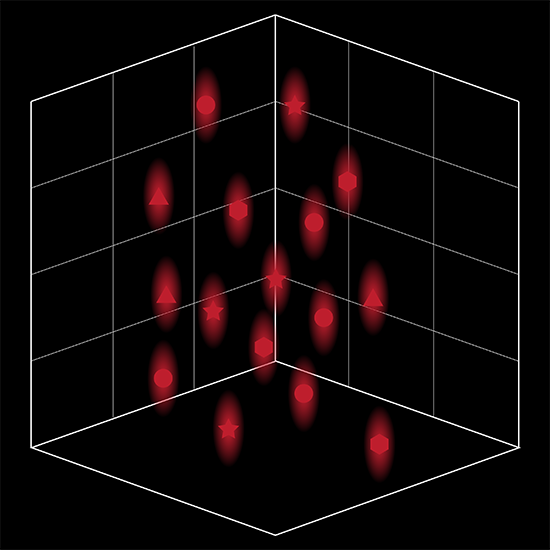
3D Holographic Stimulation
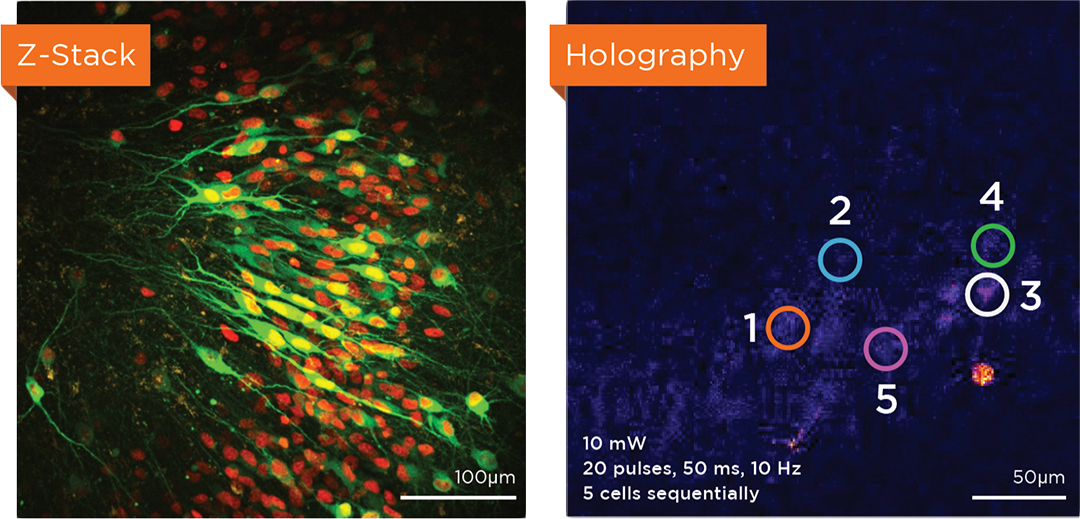
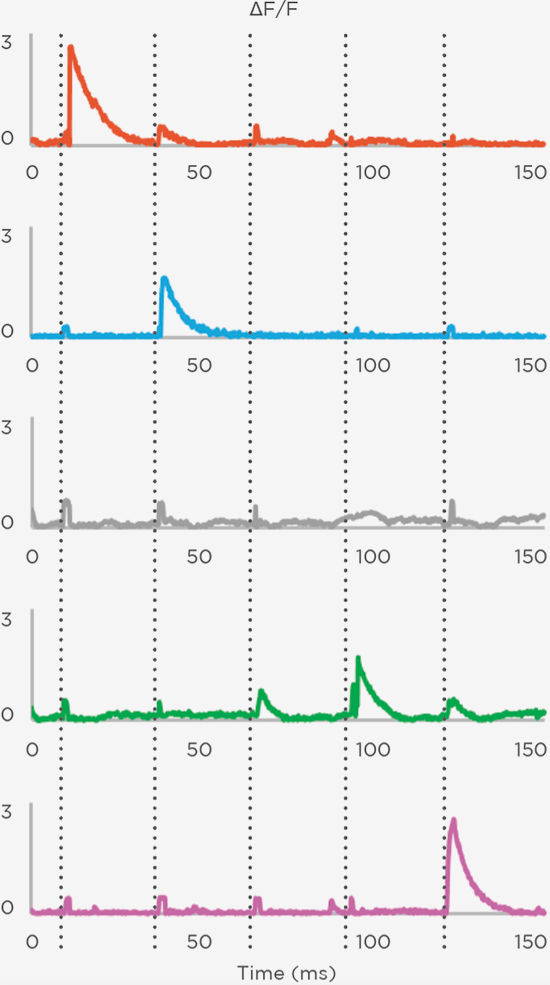
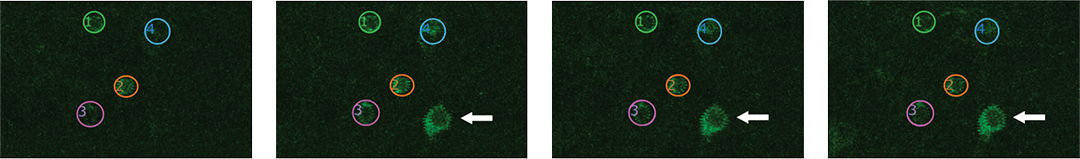
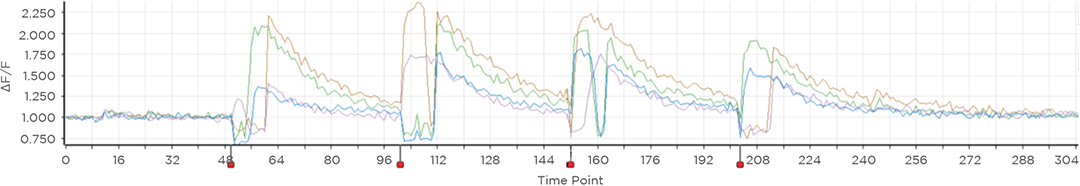
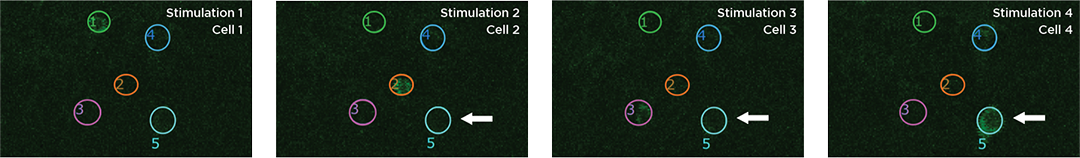
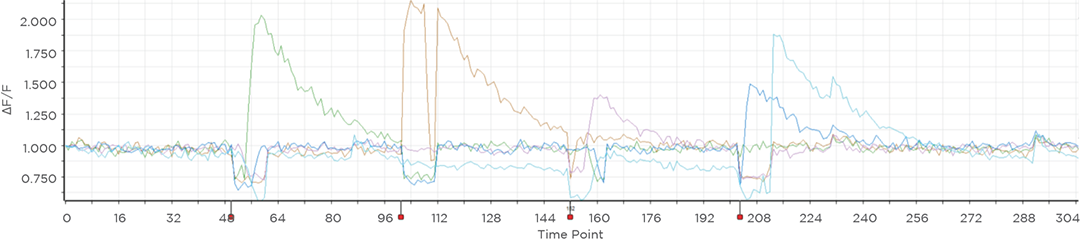
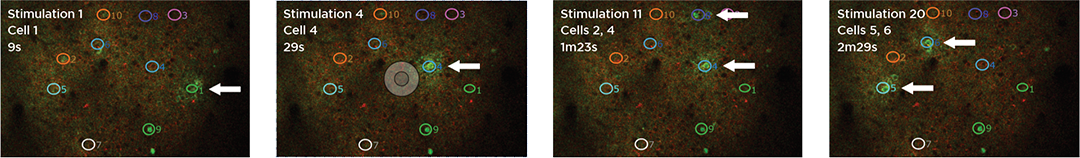
Proprietary software based on iterative Fourier transform algorithms (IFTA) simultaneously generate multiple hand-drawn regions of interest at any position in sample space.
SlideBook’s exclusively licensed libraries generate holographic patterns at multiple Z positions with corrections for power discrepancies inherent to the use of SLMs and each pattern can be assigned a specific power density.